Focus of research: AG Lars Nitschke
The group of Professor Lars Nitschke is interested in proteins of the Siglec family on immune cells. These Siglecs inhibit B cell receptor signaling and prevent development of autoimmunity and leukemia.

Prof. Dr. Lars Nitschke
Professur für Genetik
Kontakt
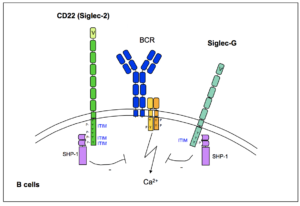
Fig. 1: The functions of CD22 and Siglec-G in the modulation of BCR-mediated signalling.
B lymphocytes (or B cells) are cells of the adaptive immune system, which induce an antibody-mediated humoral immune response. We are interested in the regulation of development and function of B cells. CD22 (Siglec-2) and Siglec-G are B cell transmembrane proteins that are associated with the B cell receptor (BCR) and interfere with its signalling cascade (Fig. 1). Both carry inhibitory ITIM motifs, which are phosphorylated after stimulation of the BCR. After phosphorylation, the ITIM motif recruits phosphatases such as SHP-1, which negatively regulates signalling. We generated CD22-/- and Siglec-G-/- mice and could show that these mice exhibit greatly increased BCR signalling, as measured through Ca2+ influx into the B cell. CD22 inhibits BCR signalling in conventional B cells, while Siglec-G inhibits BCR signalling in the subpopulation of B1 cells, which are B cells with specific functions.Both Siglec-G-deficient, as well as CD22 x Siglec-G-double deficient mice develop an autoimmune disease which resembles systemic lupus erythematosus (SLE), characterized by autoantibodies and kidney pathology. CD22 binds to specific sialic acid (Sia) containing ligands, mainly in cis, on the same cellular surface and this leads to a regulation of BCR signaling. During an immune response a germinal center (GC) forms where B cells divide and produce antibodies with better antigen binding properties. In GC B cells high affinity ligands for CD22 are downregulated. This leads to impaired BCR signaling in GC B cells. As a consequence CD22-deficient GC B cells are not maintained as well as wild type GC B cells and are more likely to die by apoptosis. The influence of lower and higher GC B cell signaling on B cell fate and metabolism is studied in a FOR5560 project.
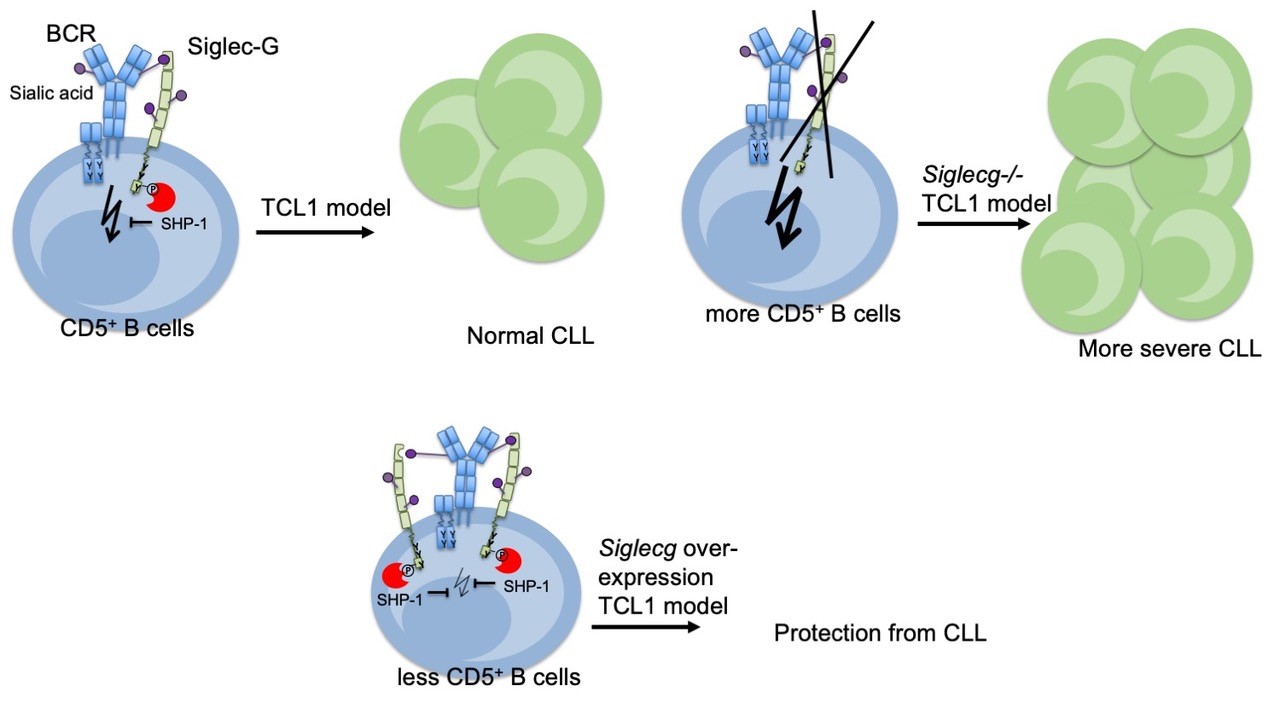
Chronic lymphocytic leukemia (CLL) is the most common leukemia in adults in the Western world. BCR signaling is known to be crucial for the pathogenesis and maintenance of CLL cells which develop from mature CD5+ B cells. Siglec-G-deficient mice have an enlarged CD5+ B1a population. Our research showed that SiglecG-deficiency leads to an earlier onset and more severe cause of CLL in the murine Emu-TCL1 model. In contrast, mice overexpressing Siglec-G on the B cell surface are almost completely protected from developing CLL disease. These results demonstrated a critical role for Siglec-G in CLL and suggest that it can be addressed as a new target (Fig.2). The mechanism how Siglec-G or CD22 regulate CLL is further studied in a Deutsche Krebshilfe project.
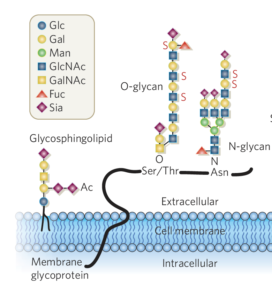
Sialic acids (Sia) are abundant carbohydrate structures on glycoproteins of cell membranes. On these glycoproteins Sia are the terminally expressed carbohydrates (Fig.3). Sia serve as ligands for Siglec proteins, regulating their inhibitory functions. We generated mice lacking Sia either on B cells or T cells. Surprisingly, B cells and T cells without Sia cannot survive in immunological organs such as the spleen or lymph nodes. This points to a more general role of Sia than just serving as Siglec ligands on lymphocytes. These functions are addressed in our group in detail. This project is funded by the FOR 2953.
Lars Nitschke on Google Scholar
5 Key Publications
- Meyer SJ,Linder AT, Brandl C, Nitschke L. (2018) B Cell Siglecs-Newson Signaling and Its Interplay With Ligand Binding. Front Immunol. 9:2820.
- Röder B, Fahnenstiel H, Schäfer S, Budeus B, Dampmann M, Eichhorn M, Angermüller A, Brost C, Winkler TH, Seifert M, Nitschke L. (2023) The inhibitory receptor Siglec-G controls the severity of chronic lymphocytic leukemia. EMBO Rep. 24(8):e56420
- Linder AT, Schmidt M, Hitschfel J, Abeln M, Schneider P, Gerardy-Schahn R, Münster-Kühnel AK, Nitschke L. (2022) Sialic acids on B cells are crucial for their survival and provide protection against apoptosis. Proc Natl Acad Sci U S A. 119(25):e2201129119.
- Schmitt H, Sell S, Koch J, Seefried M, Sonnewald S, Daniel C, Winkler TH, Nitschke L. (2016) Siglec-H protects from virus-triggered severe systemic autoimmunity. J Exp Med. 213:1627-44
- Müller J, Obermeier I, Wöhner M, Brandl C, Mrotzek S, Angermüller S, Maity PC, Reth M, Nitschke L. (2013) CD22 ligand-binding and signaling domains reciprocally regulate B-cell Ca2+ signaling. Proc Natl Acad Sci U S A. 110:12402-7
Click here for the full list of publications
Research of the Nitschke group is funded in the context of the following projects:
- DFG RTG2599: The role of di-sialic acids in plasma cell function (2024-2026)
- DFG FOR2953/P02: The importance of sialoglycans for lymphocyte development (2022-2027)
- DFG FOR5560/P03: The role of BCR signaling stength in germinal center B cell fate and metabolism (2023-2027)
- Deutsche Krebshilfe: The function of Siglecs in Chronic Lymphocytic Leukemia (2025-2028)

